Hi readers,
This week, we’re taking a deeper dive into one of the more dramatic turnaround stories in feline medicine.
For decades, feline infectious peritonitis (FIP) was one of the most devastating diagnoses for cats, with mortality rates of around 90-95% or even higher. Treatment options for FIP, an immune-mediated disease, were basically zero.
Things began to change in the late 2010s, when research showed that compounds related to the antiviral GS-441524 was effective against FIP. Despite this, the treatment was not approved for veterinary use.
Desperate to save their cats, cat guardians subsequently created a global grey market for unlicensed versions of GS-441524 made in China.
Today, things are shifting again. In this week’s Feline Business Brief, we examine how the FIP treatment landscape is evolving, and what it could mean for the future of feline medicine.
What we’re watching
🐈⬛ CATalyst Council’s survey shows 90% of devoted cat owners can increase veterinary practice revenue
How treatments for feline infectious peritonitis moved from the black market to the clinic

For decades, feline infectious peritonitis (FIP) was considered fatal.
But in less than a decade, the disease has become a striking case study of feline therapeutic innovation.
Not only that: the first effective FIP antivirals emerged in a global grey market created by cat guardians, not within any traditional veterinary pathway.
This raises the question: are we seeing the emergence of a wider feline antiviral market?
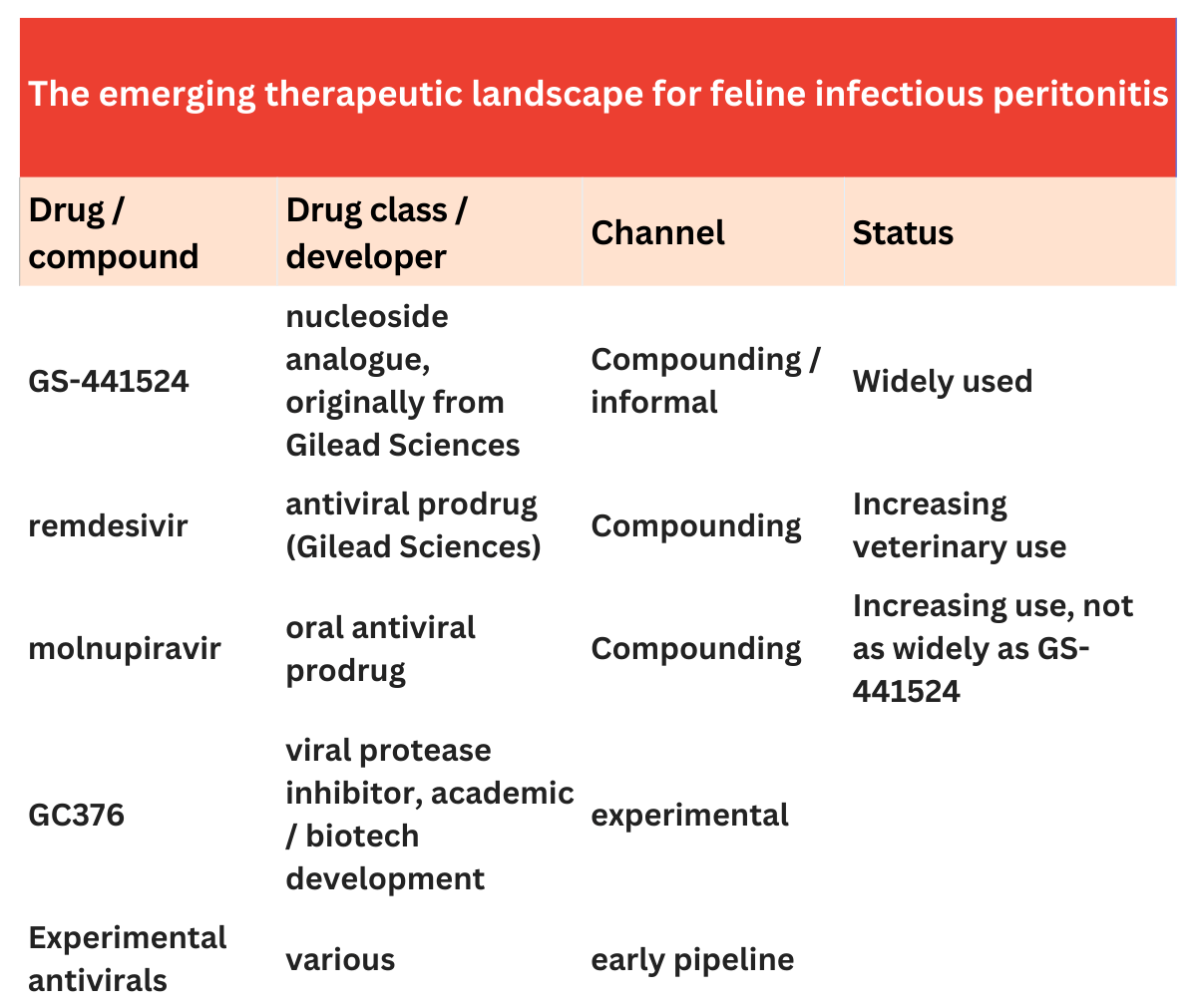
From fatal disease to treatable infection
FIP is caused by a mutated form of feline coronavirus that triggers systemic inflammatory disease. Treatment options traditionally involved supportive care, and mortality rates exceeded 90-95%.
This changed after data was published in 2018 demonstrating the efficacy of nucleoside analogue antivirals (including GS-441524) against viral replication. The research suggested survival rates of approximately 70–90% in treated cats.
Interestingly, GS-441524 is a close relation to Gilead Sciences’ remdesivir, which was later used to treat COVID-19 in humans.

An unusual path to treatment
This scientific breakthrough did not immediately lead to an approved FIP treatment.
Despite data published in 2018 from the Pedersen group work at UC Davis demonstrating GS-441524’s efficacy against FIP, Gilead Sciences declined to license the drug for veterinary use.
This sparked an extraordinary concerted global response: cat guardians across the world collaborated to find a workaround.
Cat guardians, desperate to save their cats, formed online communities to share information. The most influential the FIP Warriors Facebook group, where cat guardians shared information on how to source and administer GS-441524;
Meanwhile, pharma manufacturers in China became aware of the demand for GS-441524, and began producing unlicensed versions of the drug.
The result was the creation of a global parallel market for unlicensed GS-441524 for cats with FIP.
The black market for unlicensed GS-441524 evolves
Over time, the market for unlicensed GS-441524 started to evolve.
Veterinarians became increasingly involved in managing treatment, in the face of growing clinical evidence that GS-441524 worked against FIP. Meanwhile, regulators in several countries began signalling greater leeway for the veterinary use of antiviral compounds.
The turning point: In May 2024, the FDA published new guidelines stating that it would not prioritise enforcement against compounded GS-441524. Pharmaceutical compounding is the preparation of customised medications by pharmacists who mix, alter or combine ingredients for a specific patient’s needs, when a commercially approved drug is unavailable or unsuitable.
This enabled veterinarians to access approved drug formulations through compounding pharmacies. Similar pathways also emerged in markets such as the UK, Canada and Australia.
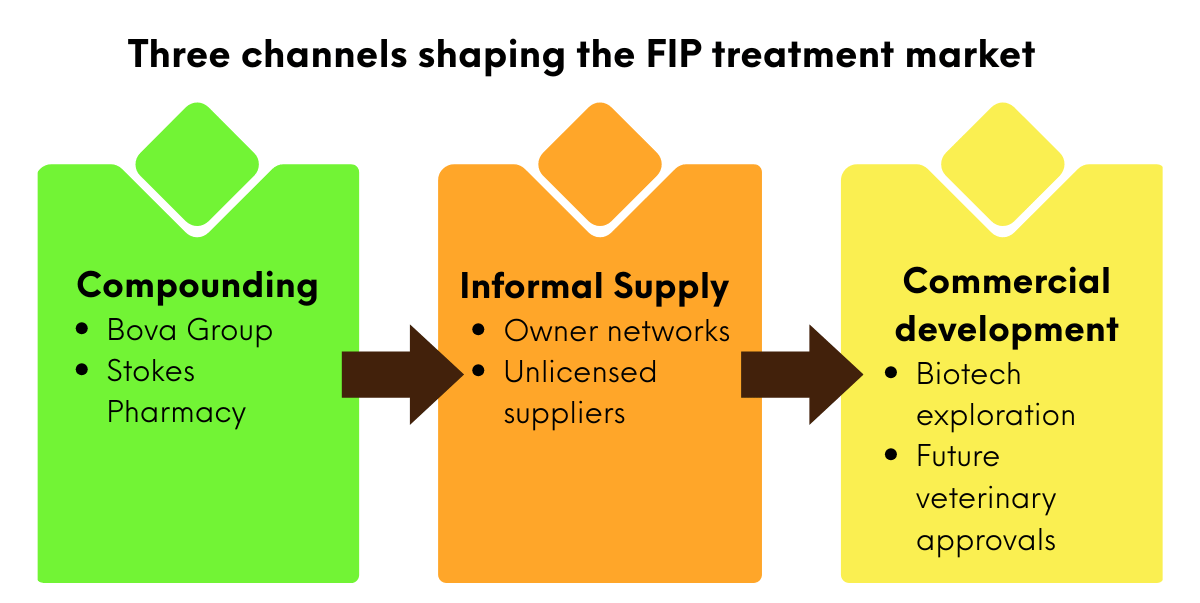
The FIP treatment landscape is currently divided into three channels:
Compounded antivirals: Compounded formulations have played a crucial role in making antivirals available to treat cats with FIP. In several countries, veterinarians now access GS-441524 or remdesivir through licensed compounding pharmacies such as Bova Group and Stokes Pharmacy. However, compounding is unable to be carried out on a large scale, which means the bottleneck to wider access to FIP treatments remain regulatory constraints.
Informal and grey-market markets: Before compounding became widely available, many cat guardians acquired GS-441524 through informal networks and unlicensed suppliers. These channels still exist, but are shrinking as legal channels and access expand.
Emerging commercialisation efforts: Several companies are exploring formal development pathways for FIP antivirals, including Anivive Lifesciences, which has investigated protease inhibitor therapies targeting FIP.
What the emerging FIP market means for the feline health space

The FIP grey market may be a prelude to the first modern antiviral treatment category for cats. This is particularly noteworthy as:
Demand for FIP treatment clearly already exists. Unlike some animal therapeutics where demand needs to be demonstrated before clinical adoption, there is clearly already strong demand and willingness to pay for FIP antivirals.
Regulatory frameworks are evolving. The FDA condoned the use of compounding to treat FIP, which may indicate further flexibility later on, potentially leading to full regulatory approval.
These factors also highlight a recurring characteristic of feline medicine: innovation is often driven from the bottom up by cat guardians using real world evidence, rather than by pharmaceutical companies or regulators.
In the case of FIP, cat guardians effectively built a parallel pharmaceutical market and then pressured regulators to respond.
Is an ‘official’ FIP market forming?
There are several developments that indicate that a structured market is forming for FIP treatment. This is driven by:
Proof of disease mechanism: The viral replication pathways of FIP are now much better understood;
Coronavirus research spillover: The COVID-19 pandemic accelerated the development of mRNA and coronavirus vaccines;
Growing feline health investment: the cat sector is attracting more R&D funding overall;
Regulatory pathways have become more flexible: Regulatory agencies in several major markets have become more lenient towards the use of FIP treatments.
An interesting development: treatment first, then vaccine
In most cases, vaccines against infectious diseases usually appear before treatments. In the case of FIP, the reverse seems to be true, with treatments appearing before vaccines.
That said, global R&D into FIP vaccines is accelerating:
Zoetis’ Vanguard Feline FIP Intranasal is currently the only approved feline FIP vaccine;
Academic groups in the US and Europe are researching novel vaccine approaches, including mRNA platforms targeting feline coronavirus proteins;
In China, some drugmakers are already advancing mRNA vaccine candidates toward clinical trials. In July 2025, Jinyu Baoling Biopharmaceutical received clinical trial approval from China’s Ministry of Agriculture and Rural Affairs for its FIP mRNA vaccine. Other Chinese companies working on mRNA vaccines against FIP include Changzhou Fubang Biopharmaceutical, Ruipu Bio, and Sichuan Jixing Animal Pharmaceutical.
Looking ahead: Will this lead to the first major feline antiviral market?
If antivirals attain formal regulatory approval, FIP could become one of the first cases where feline medicine has shifted so quickly from limited options to a structured pharmaceutical market.
In turn, the success of antivirals may spur further innovation in feline infectious disease. A lot depends, however, on regulators’ willingness to approve feline-specific antivirals, as well as pharma industry appetite to develop them.
When cat guardians drive therapeutic innovation

A typically feline route to market.
The story behind the creation of a market for FIP antivirals is striking, but not altogether unusual in the feline space, where cat guardians (rather than pharma companies or regulators) are the drivers of therapeutic innovation.
While there are few other treatments that match the speed and scale of the FIP market, there are other cases where cat guardians have been key in accelerating therapeutic development:
Hypertrophic cardiomyopathy (HCM): HCM is the most common heart disease in cats, and for decades has lacked disease-modifying treatments. However, feline breed-focused groups, especially those with Maine Coon and Ragdoll cat guardians, have helped fund research into potential therapies, including sirolimus (rapamycin).
In 2025, the FDA granted conditional approval to TriviumVet’s Felycin-CA1 (sirolimus delayed-release tablets) for the treatment of subclinical HCM in cats.
Feline diabetes: Cat guardian communities, such as the Feline Diabetes Message Board, popularised home glucose monitoring using human glucometers and insulin protocols. Subsequent studies confirmed that cat guardians can reliably perform home monitoring outside the clinic.
Breed-specific diseases: Cat guardians have also helped to fund genetic research into breed-specific diseases, such as polycystic kidney disease in Persians.
These cases highlight a clear dynamic in feline medicine.
While pharmaceutical innovation in dogs often follows a traditional top-down model led by industry, progress in cats has sometimes emerged from grassroots demand.
The FIP antiviral story represents the most dramatic example to date; this indicates that engaged cat guardian groups can sometimes be seen as early signals of future therapeutic markets.
Feline Business Brief provides competitive intelligence on the global cat sector. We analyse early signals, emerging risks and structural shifts across feline health, therapeutics, diagnostics, nutrition and technology.
Feline Business Brief provides:
Global feline pipeline tracking
Market sizing
Competitive landscape mapping
Bespoke research
